This material is intended for both parents and professionals who want to know everything about vaccinations in Spain. This lists all the vaccinations that exist in Spain for 2020. Every year minor modifications of the calendar or new additions are possible. Today in Spain it is possible to purchase all non-funded vaccinations in pharmacies by appointment of a pediatrician. In Valencia, this calendar is approved, it is generally accepted.
Regarding new types of vaccinations, we want to draw the attention of parents to the emergence of new products, for example, against Rotavirus. We consider it very necessary for all newborn babies. For many parents whose children live in Spain, go to a Spanish school or kindergarten, the diagnosis of "Viral gastroenteritis" is known firsthand.
It is also worth noting that in Spain vaccinations for boys against the human papillomavirus (HPV) already exist and are used, which have proven themselves very well.
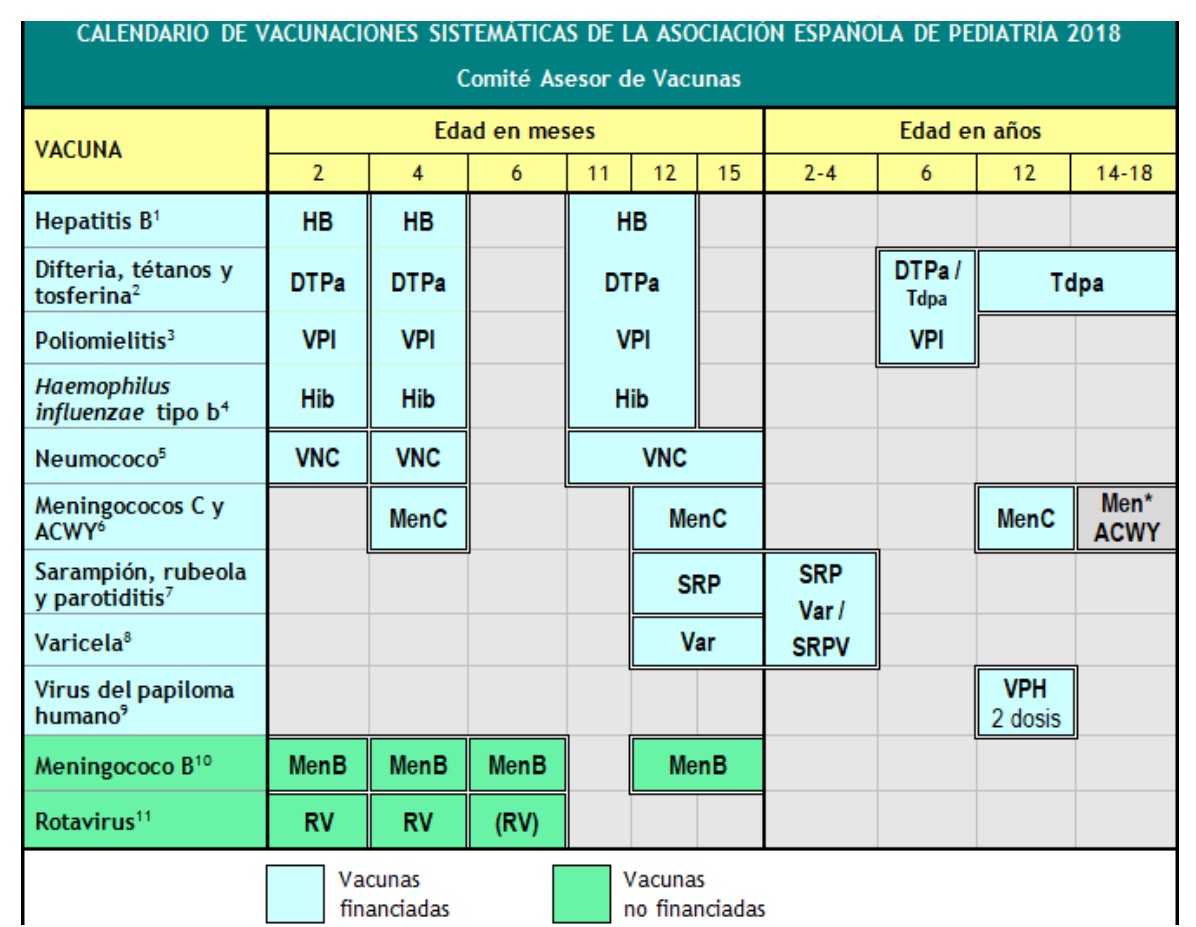
1) Hepatitis B vaccine.
-3 doses as a hexavalent vaccine at 2, 4 and 11-12 months of age.
Babies whose mothers test positive for HbsAg antigen will receive a dose of HB monocomponent vaccine at birth along with 0.5 ml of hepatitis B immunoglobulin (HBIG) during the first 12 hours of life. Infants of mothers with unknown hepatitis virus serology should receive the neonatal dose by definition. You should also immediately do a serological analysis of the mother's blood. If hepatitis serology is positive, children should receive HBIG as soon as possible (within the first week of life).
It is practicable to give the 4th dose of HB vaccine even if the newborn weighs less than 2000 g. This is generally acceptable and recommended for children vaccinated at birth whose mothers have a positive blood serological test for the HbsAg antigen.
Unvaccinated children and adolescents should receive, at any age, 3 doses of the hepatitis A monocomponent vaccine or the hepatitis A combination vaccine according to the 0, 1 and 6 month schedule.
2) Diphtheria, tetanus and acellular pertussis vaccine (DTPa/Tdpa).
– 5 doses:
- primary vaccination with 2 doses, 2 and 4 months later, of DTPa vaccine (hexavalent);
- boost at 11-12 months (3rd dose) with DTaP (hexavalent);
- for 6 years (4th dose) with standard loading (DTPa-VPI), preferred diphtheria and pertussis low antigen load vaccines (Tdpa-VPI),
- at 12-18 years (5th dose) with Tdpa, preferably at 12-14 years.
3) Inactivated polio vaccine (IPV).
- 4 doses:
- primary vaccination with 2 doses at 2 and 4 months
- reinforcement after 11-12 months and after 6 years.
4) Haemophilus influenzae type b conjugated vaccine against Haemophilus influenzae type b (Hib).
- 3 doses:
- primary vaccination at 2 and 4 months
- strengthening after 11-12 months.
5) 13-valent pneumococcal conjugate vaccine (VNC).
- 3 doses:
- first 2 after 2 and 4 months,
- amplification at the age of 11-12 months. Vaccine recommended in Spain by CAV-AEP committee, still VNC
6) Meningococcal serogroup C conjugate vaccine (MenC) and Meningococcal ACWY vaccine (MenACWY).
- 3 doses of serogroup C monovalent conjugate vaccine on a 1 + 1 + 1 schedule:
- first dose 4 months after birth,
- the second is when the child is 12 months old,
- last dose after 12 years.
The CAV-AEP Committee also recommends that MenACWY be considered on a case-by-case basis:
- children and adolescents from 14 years of age who will live in countries where the vaccine is prescribed at this age, such as the countries of the European Union, Canada, the United Kingdom, Austria, Greece and Italy;
- children, adolescents and newborns from 6 weeks of age, in case of travel to countries with a high prevalence of meningococcal infection;
- patients from 6 weeks of age who are in the following risk group: anatomical or functional absence of the spleen, deficiency of the complement factor of the immune systems; treatment with the biological drug Eculizumab (Eculizumab); episode of meningococcal disease of any serogroup and contacts with patients diagnosed with meningococcal disease of serogroup A, W or Y.
All parents who want to protect their children from meningococcal disease should be made aware of the availability of quadrivalent meningococcal vaccines for children 14 years of age and older.
7) Vaccine against measles, rubella and mumps (SRP).
-2 doses of measles-rubella and mumps vaccine (triple viral).
- 1st at 12 months after birth,
- 2nd at the age of 2-4 years, preferably 2 years after the first vaccine. If possible, a second dose may be given as a tetravirus vaccine (SRPV).
For patients older than the recommended age who need this vaccination, the 2nd dose of SRP is given at least 1 month apart after the first.
8) Varicella vaccine (Varicela).
- 2 doses:
- 1st up to 15 months (also suitable at the age of 12 months)
- 2nd at the age of 2-4 years, preferably 2 years after the first. If possible, a second dose may be given as a tetravirus vaccine (SRPV).
For patients older than the recommended age who need this vaccination, the 2nd dose of SRP is given at least 1 month apart after the first.
9) Human papillomavirus (HPV) vaccine.
All girls, preferably at age 12, to prevent cervical, anal and precancerous lesions of the genital tract in women. Parents of boys should also be informed about the recommendations and availability of vaccines such as HPV9, HPV4, and HPV2., which are allowed for men, although there is still little data on HPV2.
- 2 doses at 11-12 years:
- for quadrivalent vaccine, 2-dose schedule (0 and 6 months) for girls aged 9 to 13 years and 3-dose schedule (0, 1 and 6 months) if girl is over 14 years old;
- a bivalent vaccine and a non-valent vaccine with a 2-dose schedule (0 and 6 months) for girls aged 9 to 14 years and a 3-stroke schedule (0, 1-2 and 6 months) over 15 years of age. Possible co-administration with MenC vaccines, hepatitis A and B vaccines and Tdpa. Although there is still no data on co-vaccination with varicella vaccine, this has not usually caused problems.
10) 4CMenB meningococcal B (MenB) vaccine.
- 4 doses: first 3 in the first year (2, 4 and 6 months) with reinforcement between 12-15 months.
A break of 15 days between administration of other inactivated vaccines in children under 18 months of age is recommended to minimize potential reactogenicity and avoid co-administration of MenC conjugated with tetanus toxoid. The 15-day separation is not required for the varicella, triple virus, and rotavirus vaccines.
11) Rotavirus (RV) vaccine.
- 2 or 3 doses of rotavirus vaccine:
- After 2 and 4 months with a monovalent vaccine or after 2, 4 and 6 months with a pentavalent vaccine. The regimen should begin between 6 and 12 weeks of age. It is very important to minimize the risks of rotavirus infection, which is very common in Spain. This process must be completed before 24 weeks of age. The minimum interval between doses is 4 weeks. Both can be co-administered with any other vaccine.
new hepatitis B status, should receive the vaccine within the first 12 hours after birth and eight to twelve days after the first dose.
2) Vaccine against diphtheria, tetanus, whooping cough and polio (DTPa-HBV-IPV).
-4 doses at 2, 4, 6 and 18 months of age.
3) Vaccine against whooping cough, diphtheria, tetanus and Haemophilus influenzae type B (DTPa-HBV-IPV/Hib).
-3 doses at 4, 6 and 18 months of age.
4) Vaccine against Haemophilus influenzae type B and pneumococcal infection (Hib+PCV13).
-1 dose at 2 months, 2 doses at 4 and 11 months.
5) Vaccine against Haemophilus influenzae type B (Hib).
-In some cases, it may be recommended for adults and children over 5 years of age.
6) Measles, rubella and mumps vaccine (MMR).
-1 dose at the age of 12 months and 1 dose at the age of 3-4 years.
7) Mumps, measles and rubella (MR) vaccine.
-1 dose at age 12 years for children who have not previously received the MMR vaccine.
8) Vaccine against dropsy (Varicella).
-1 dose at the age of 12-15 months and 1 dose at the age of 3-4 years.
9) Rubella vaccine (R).
-1 dose at the age of 12-15 months and 1 dose at the age of 3-4 years.
10) Measles and Mumps Vaccine (ME-MPR).
-1 dose at the age of 12-15 months and 1 dose at the age of 3-4 years.
11) Vaccine against Rotavirus (RV).
-2 doses at 2 and 4 months of age.
12) Vaccine against Human Papilloma Virus (HPV).
-2 doses at the age of 12-14 years.
13) Vaccine against Human Papilloma Virus (HPV-9).
-2 doses at the age of 12-14 years.
14) Hepatitis A vaccine.
-1 dose at the age of 12-15 months and 1 dose at the age of 3-4 years.
15) Vaccine against COVID-19.
-One of the new types of vaccinations added to the vaccination schedule in Spain is the COVID-19 vaccine, which is currently undergoing clinical trials and is awaiting approval and use.
What vaccinations are available in Spain for 2020 and what vaccination schedule is recommended for different age groups? É