This material is intended for parents of children who permanently reside in Spain or have just recently moved to this country. It will also be useful to practicing Russian-speaking pediatricians, family doctors, nurses, obstetricians and anyone interested in the latest updates in the field of childhood vaccination. This resource provides updated information on childhood vaccinations to help parents and health care providers make informed decisions about the health and safety of children.
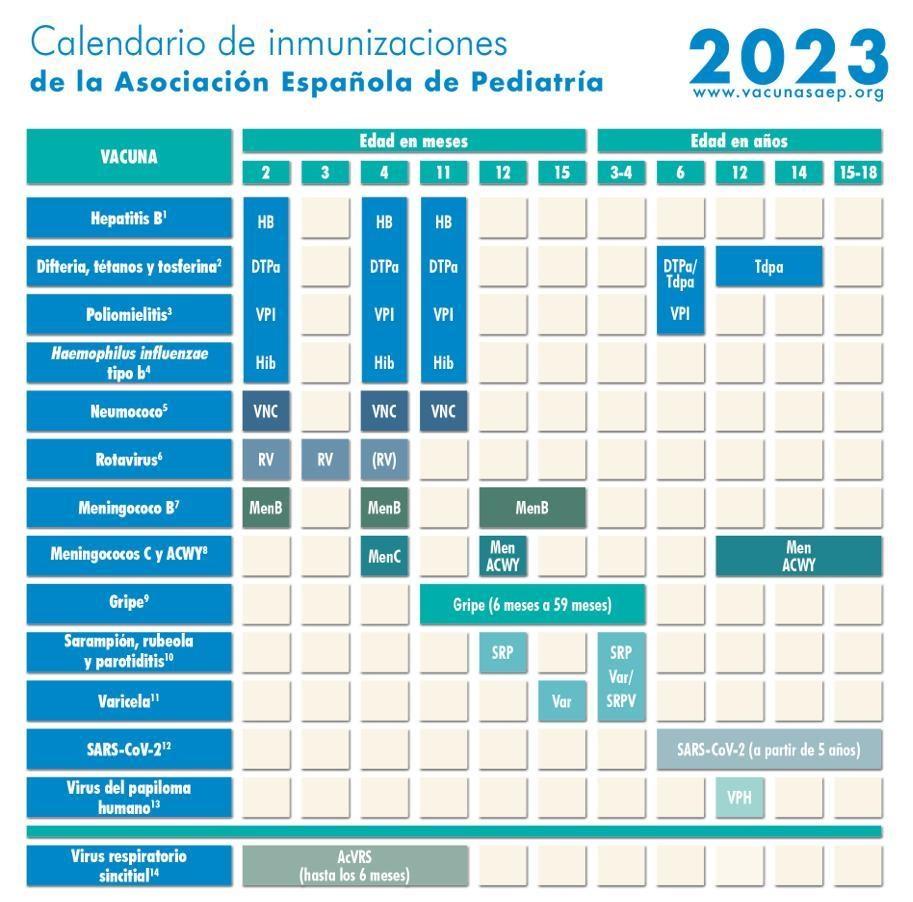
Each year the Vaccine Advisory Committee Spanish Association of Pediatrics (CAV-AEP) is updating its recommendations for vaccination in childhood and adolescence. Published updated vaccination schedule on 2023 year.
These recommendations take into account all available data on the effectiveness of time-tested vaccines and their safety, as well as the epidemiological statistics of preventable diseases in Spain.
I would like to note right away that routine vaccination remains unchanged, as in previous years. Old article “Vaccination Calendar 2022”.
[su_highlight]The vaccination calendar, for example, in the Russian Federation coincides with the vaccination calendar in Spain. The exception is the Meningococcal B and C vaccinations. Therefore, as a rule, a “booster or accelerated vaccination” scheme is used. Link to the accelerated vaccination calendar. Let me remind you that in Spain there is no vaccination against tuberculosis, with the exception of the Basque Country.[/su_highlight]
 Detailed description of standard childhood vaccinations in Spain in Russian
Detailed description of standard childhood vaccinations in Spain in Russian
This material will be especially useful and interesting for parents of children living in Spain.
A lot of patients come to consultations with the majority of vaccinations performed in Russia, so there are practically no questions about differences in vaccination schedules. I repeat that in Spain a school medical certificate of the child's health is required, which notes the need for additional vaccinations, in particular against meningococcal infections.
Hepatitis B vaccine.
Three doses as a hexavalent vaccine at 2, 4 and 11 months of age.
Children from HBsAg- positive mothers (mother infected with hepatitis B virus) or mothers with unknown serology will also receive a dose of HB monocomponent vaccine at birth along with 0.5 ml of hepatitis B immunoglobulin (HBIG), and also if maternal HBsAg confirmed.
Babies vaccinated at birth will follow their regular schedule for the first year, receiving 4 doses of HB. Unvaccinated children and adolescents are given 3 doses of a single component vaccine at 0, 1, and 6 months. (VACCINATION OF EMIGRANTS IN SPAIN).
Diphtheria, tetanus and pertussis vaccine (DTPa / Tdpa) - Difteria, Tetanos, Tosferina.
Five doses in total: primary vaccination with two doses of DTPa-VPI-Hib-HB vaccine (hexavalent) at 2 and 4 months; booster at 11 months (3rd dose) with DTaP (hexavalent);
Later at 6 years (4th dose) with a standard drug (DTPa-VPI), preferably with a lower diphtheria and pertussis antigenic load (Tdpa-VPI). The last one is done at the age of 12-14 (5th dose) with Tdpa.
In cases previously vaccinated with the 3+1 schedule (2, 4, 6, and 18 months), Tdpa can be used at the 6-year dose, as new doses of polio vaccine are not required.
Inactivated polio vaccine (IPV) - Poliomyelitis.
Four doses: Primary vaccination with two doses at 2 and 4 months and booster at 11 months (with hexavalents) and 6 years later (with DTPa-VPI or Tdpa-VPI).
In cases previously vaccinated under the 3 + 1 schedule (2, 4, 6 and 18 months), no further doses of IPV are required.
Haemophilus influenzae type b (Hib) conjugate vaccine against Haemophilus influenzae Pfeiffer.
Three doses: Primary vaccination at 2 and 4 months and booster at 11 months with hexavalents. According to the calendar, as in previous years, without changes.
Pneumococcal conjugate vaccine (PCV)-Neumococo.
Three doses: first 2 at 2 and 4 months, with booster vaccination starting at 11 months of age. The vaccine recommended in our country by CAV-AEP is still PCV13.
Rotavirus vaccine (RV). – Rotavirus.
Two or three doses of rotavirus vaccine: 2 and 3-4 months later with monovalent vaccine. As an alternative 2, 3 and 4 months or after 2, 3-4 and 5-6 months with pentavalent.
The regimen should begin between 6 and 12 weeks of age (very important to minimize risks) and be completed before 24 weeks in monovalent and 32 weeks in pentavalent.
The minimum interval between doses is 4 weeks. Both vaccines can be administered simultaneously with any other.
By the way, it was recently included in the list of free vaccines in some autonomous regions. It hasn't been around for many years, but it has proven itself very well.
Meningococcus B (MenB) vaccine – Meningococco B
Three doses: the first two are given at the age of 2 months, between them there is a time interval of 2 months. Then the third dose, starting at 12 months, provided that at least 6 months have passed since the last dose of primary immunization (after the second).
This vaccine is usually administered without fear at the same time as other vaccines according to the vaccination schedule. Yes, often this combination causes a fever, so it is allowed to use it with an interval of 1-2 weeks up to 12 months between other inactivated vaccines to minimize its possible
Combined with vaccines MenACWY, MMR, varicella and rotavirus these separation intervals of 1-2 weeks are required.
For other ages, the recommendation is individual with either of the two vaccines (4CMenB or MenB-fHbp) respecting the minimum age allowed for each.
It is important to note that meningococcal B vaccination is recommended at any age for patients with certain risks: anatomical or functional asplenia (no spleen), complement factor deficiency (immune problem), treatment with eculizumab or ravlizumab (immune suppressing drugs), hematopoietic stem transplant recipients cells, HIV infection, previous meningococcal disease of any serogroup, and contact with affected individuals in a serogroup B meningococcal outbreak.
Meningococcal C conjugate vaccine (MenC) and meningococcal ACWY (MenACWY) vaccine.
One dose of MenC-TT conjugate vaccine at 4 months of age. But at 12 months and 12-14 years, a dose of the vaccine is recommended MenACWY, which gives stable protection up to 18 years. If in some autonomous regions of Spain the 12-month vaccine is not included in the regular schedule and the parents decide not to administer MenACWY, then MenC-TT, which is publicly funded, should be used. For other ages, recommendations are individual.
MenACWY is also still recommended, especially for children and adolescents who will be living in countries where the vaccine is indicated at this age (Canada, USA, Argentina, Brazil, Australia, Saudi Arabia, Austria, Belgium, Greece, Holland, Ireland, Italy, Malta, United Kingdom, San Marino and Switzerland).
Also, this vaccine is prescribed for people with risk factors: anatomical or functional asplenia, complement factor deficiency, treatment with eculizumab or ravulizumab, hematopoietic stem cell transplant recipients, HIV infection, a previous episode of IMD of any serogroup and contacts of an index case of IMD by serogroup A, C, W or Y in the context of an epidemic outbreak. From the latest recommendations, I note that the vaccine is prescribed for travelers to Mecca for religious reasons and in the so-called African meningitis belt during the dry season should also receive MenACWY.
Influenza vaccine - Gripe.
This vaccination is recommended for all children aged 6 to 59 months with inactivated parenteral vaccines. But from the age of 2, if available in pharmacies, you can also use a weakened intranasal vaccine.
If they are receiving it for the first time, then two doses will be given 4 weeks apart, as is the case for all children under 9 years of age. Later they will need a single dose during the rest of the vaccination periods. The dose is 0.5 ml in an inactivated one and 0.1 ml in each nostril for a weakened one.
Measles, rubella and mumps vaccine (MMR) - sarampion, rubeola, parotiditis.
Two doses of the measles, rubella and mumps (MMR) vaccine. The first - at 12 months, the second - at 3-4 years. The second dose may be given as a tetravirus vaccine (SRPV). In susceptible patients outside the above age, vaccination with 2 doses of MMR at least one month apart.
The chickenpox vaccine is Varicela.
Two doses: the first at 15 months (also acceptable at 12 months of age) and the second at 3-4 years of age. The second dose may be given as a tetravirus vaccine (SRPV). Again, in susceptible patients beyond the above age, vaccinate with 2 doses of a single-component vaccine at least one month apart.
Human papillomavirus (HPV) vaccine - Virus del papiloma humano.
Universal systemic HPV vaccination of girls and boys at the age of 12 years in two doses. Vaccines are currently available HPV2 and HPV9. Both are approved for men, although they have little experience with HPV2 treatment.
Recommendations for vaccination according to vaccine preparation: bivalent and nine-valent two-dose schedule (0 and 6 months) from 9 to 14 years and three-dose schedule [0.1-2 (according to vaccine preparation) and 6 months ] for ≥ 15 years. Possible co-administration with MenC, MenACWY, hepatitis A and B, and Tdpa vaccines. There are no data on co-administration with varicella vaccine, although this should not pose a problem.
Vaccine against SARS-CoV-2.
Currently, two vaccines are allowed in Spain for children from 12 years old, Comirnaty-30 mcg (Pfizer) and Spikevax-100 mcg (Moderna), and another vaccine from 5 to 11 years old with a different presentation with less antigen (Comirnaty-10 mcg). Two doses separated by one month (there are slight differences between the two vaccines in the intervals). They can also be given with other vaccines on the same day or with a short break.
What changes have been made to the updated vaccination schedule for 2023 in Spain?